Regulatory Intelligence and Workflows, Right Where You Work: Introducing Formly's MCP Server
Formly now has an MCP server. Connect your Formly workspace directly to Claude or GPT and run regulatory research, standards lookup, and expert review without switching tools.
Every piece of compliance software on the market shares the same assumptions:
- that you'll come to it.
- that you bring the necessary expertise
Log in. Navigate the dashboard. Find the tool you need. Work its way. That's been the implicit contract with eQMS software for twenty years, and most of the industry hasn't questioned it. We have.
That's why as of today Formly has its own MCP server. That means you can connect Claude or ChatGPT directly to your Formly workspace and access our regulatory intelligence, expert agents, and your compliance data without ever leaving the AI environment where you already work. We built Formly to come to you. Not the other way around.
Two problems we've been thinking about
The context-switching tax
Here's something that's become true of how regulatory work actually gets done: a lot of it starts in Claude or ChatGPT. Predicate research begins as a conversation. Standards lookup happens in a chat window. Document drafts get workshopped with an AI before they go anywhere near a formal system.
And we see that trend accelerating. AI tools are increasingly the place for people to work on tasks or whole projects holistically. But in a regulated industry like for medical devices eventually the moment comes where you need real regulatory infrastructure. A proper predicate comparison, reference check for a testing strategy against standards that goes beyond Googling it, a document review that applies actual compliance criteria and depends on years of industry experience or a simple document that needs to be routed for approval so it is stored compliant with ISO 13485. So you have to stop. Open another tab. Log into another tool. Call or email your consultant. Context switch.
That interruption is a tax. It's not one big cost, it's a hundred small ones across a working week. And it compounds for anyone doing regulatory work at volume: the founder who does it themselves, consultants managing multiple clients, startup RA leads switching between submissions, quality teams reviewing documents in parallel. The tools should follow the work, not the other way around.
The expertise access problem
There's a second issue that's less obvious but just as significant.
Until now, accessing expert-level regulatory intelligence has required one of two things: hiring talent, or a consultant. A regulatory strategy, a predicate research for the next Pre-Sub that actually holds up, standards review that covers the right frameworks, document review that applies a trained regulatory lens. These capabilities have always been gated. Behind onboarding. Behind contracts. Behind cost.
When you connect the Formly MCP server to Claude or ChatGPT, both of those requirements go away. You get direct access to Formly's regulatory knowledge base and expert agents. The same intelligence that powers us and our platform as tools your AI can call the moment you connect.
That's a different kind of value proposition existing software in the industry offer. It's not a feature update but a different relationship with the knowledge and tools you need.
What actually is an MCP ?
Most of you will know this but its worth reiterating once in a while. MCP stands for Model Context Protocol. An open standard, developed by Anthropic, that defines how AI assistants connect to external tools and data sources. Think of it as the integration layer that lets a model like Claude go beyond its training data and actually interact with live systems: read documents, call APIs, trigger workflows, and return structured results.
The MCP ecosystem has grown quickly in the developer and productivity space. There are MCP servers for databases, version control systems, communication tools, and cloud platforms. Regulated industries, though, have been an obvious gap. And it's an especially significant one, because compliance work is exactly the kind of task that benefits most from this architecture. It's deeply context-dependent. The right answer depends on your specific device, your market, your submission pathway. It's documentation-heavy. And it's expert-knowledge-intensive in a way that makes it genuinely hard to do well without domain-specific intelligence behind the AI.
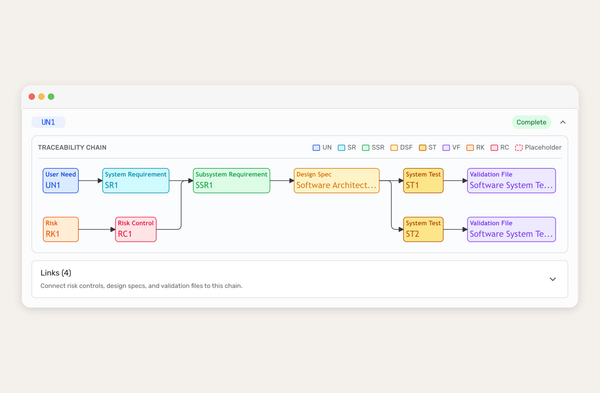
Formly already knows your medical device, now Claude does too.
The moment you connect the Formly MCP server, Claude has access to everything your workspace already holds. Your intended use statement, your device classification, your target markets, risk classification, and predicate references. Not as context you paste in at the start of every session but as structured data keys it can retrieve precisely, on demand, every time.
That distinction matters. When you or your AI ask "what's our intended use?", Claude doesn't infer or approximate based on what you've described. It retrieves the exact text from your QMS. Ask it to create a risk management file from your template, and that same text flows straight in, alongside every other data key your workspace holds. The document is structured correctly and pre-populated with your actual information. You edit and refine. You don't start from a blank page.
The same applies to the operational steps that follow. Route a document for approval, assign a reviewer, check the status of an open workflow. All from the same conversation. Research, draft, route. One session, no context switch.
It's an art, not a science
This is where the Formly MCP server becomes genuinely different from a standard integration. And different from just using Claude or ChatGPT alone for regulatory work.
What we've exposed through the MCP server isn't generic AI capability applied to compliance. It's Formly's actual regulatory expertise, encoded into agents that any AI client can invoke directly. The knowledge base behind these agents has been built and maintained by regulatory specialists. The review criteria the Expert Reviewer applies are drawn from real submission experience, not scraped from public documentation. The standards and guidances library the researcher uses is curated and grounded by citations, not a keyword index of whatever happens to be on the internet.
That’s a key difference. There's no shortage of ways to ask Claude about FDA regulations. What's been missing is a way to give Claude access to reliable regulatory intelligence that's actually been validated in the field. And to do it without a contract, an onboarding process, or a context switch out of the workflow where you're already thinking.
These three agents are how we're starting.
Predicate Researcher
Finding qualified predicates for a 510(k) submission is one of the most time-intensive early steps in any US regulatory pathway. The standard approach involves hours in the FDA's 510(k) database, searching by product code, filtering by device type and cleared date, reading through predicate summaries one by one, cross-referencing indications for use, checking technological characteristics. For a consultant working across multiple clients, this process repeats constantly and at significant cost.
The Predicate Researcher agent replaces that workflow. Describe your device, what it is, what it does, how it's intended to be used, and the agent searches the FDA database and returns qualified predicates with summaries, directly in your Claude conversation. What used to take days or weeks takes minutes. And because the results come back inside your AI workflow, you can immediately start building on them: drafting your predicate comparison, developing your substantial equivalence argument, exploring alternative pathways. Without leaving the conversation or opening another tool.
Standards & Guidances Researcher
Knowing which regulatory standards apply to your specific device is harder than it sounds, and the difficulty is rarely about effort. It's about experience. It's an art, not a science. The landscape spans ISO, IEC, FDA guidance documents, harmonised standards under MDR and IVDR, and an evolving set of regional variations. What's applicable depends on your device class, intended use, target market, and risk profile. Getting it right requires a kind of pattern recognition that takes years to develop.
The Standards & Guidances Researcher gives you that pattern recognition on demand. It searches Formly's curated regulatory standards library semantically. We’re not talking keyword matching against a static list, but meaning-based retrieval that surfaces relevant standards even when you don't know the exact title or citation. But it goes even one step further. It reasons on top of the results in an iterative loop and looks at the task at hand from multiple angles to ultimately deliver you the standards and guidances grounded answer you need with confidence. Ask about bio-compatibility requirements for a Class II wearable, or post-market surveillance obligations under MDR, and the agent returns the standards and guidance sections that apply to your situation.
For teams, this changes what's possible at the early stages of a project. You're no longer dependent on having someone in the room who already knows the standards landscape. That knowledge is accessible as a tool.
Expert Reviewer
This is the agent with the highest ceiling. And the one that replaces the most expensive step in the regulatory workflow.
Document review. Applying a trained regulatory lens to a technical file, a clinical evaluation report, a risk management file, or a design history file is expensive because it requires expertise that takes years or decades to develop. A reviewer needs to know not just what the document says, but whether it says the right things, in the right structure, with the right evidence behind its claims. That's why it's typically done by senior consultants, at significant cost, late in the project timeline when changes are hardest to make.
The Expert Reviewer agent brings that capability earlier and makes it more accessible. Push a document to the agent, and it reviews it against compliance criteria and dedicated knowledge and returns structured feedback. Not grammar suggestions or formatting notes, but a substantive assessment from a regulatory perspective.
Used in combination, these three agents cover a significant share of what regulatory teams spend their time on.
Connect in under five minutes
The setup is intentionally simple.
- Open Claude Desktop (or any MCP-compatible client) and go to settings
- Add the Formly server URL: https://mcp.formly.ai/mcp
- Set the transport to Streamable HTTP (or HTTP/SSE)
- Authenticate via OAuth. Click 'connect', a browser window opens, you sign in, done.
- Restart your client if needed
That's it. Full setup instructions are at docs.formly.ai/formly-for-ai/mcp-server.
One note: the MCP server is currently experimental. The API may change as we learn and iterate. If you run into anything, we want to hear about it.
Try it
Connect the Formly MCP server to Claude today: docs.formly.ai/formly-for-ai/mcp-server
If you're a regulatory consultant, a medtech team, or a developer evaluating how to add regulatory intelligence to an AI workflow. Try it and tell us what you find. We're actively iterating and feedback at this stage shapes what gets built.